 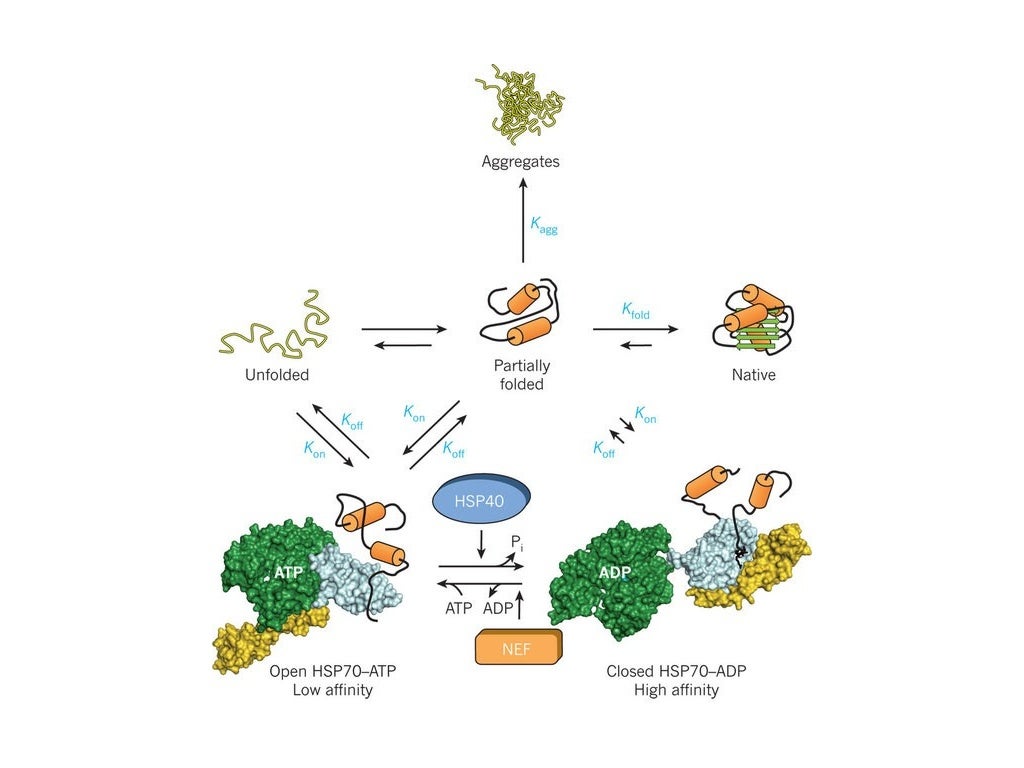 
As newly synthesized MHC class I α-chains enter the endoplasmic reticulum, calnexin binds on to them retaining them in a partly folded state. Ĭalnexin associates with the protein folding enzyme ERp57 to catalyze glycoprotein specific disulfide bond formation and also functions as a chaperone for the folding of MHC class I α-chain in the membrane of the ER. Yos-9 recognizes mannose residues exposed after α-mannosidase removal of an outer mannose of misfolded glycoproteins. The mannose lectin Yos-9 (OS-9 in humans) marks and sorts misfolded glycoproteins for degradation. The improperly-folded glycoprotein chain thus loiters in the ER and the expression of EDEM/Htm1p which eventually sentences the underperforming glycoprotein to degradation by removing one of the nine mannose residues. If the glycoprotein is not properly folded, an enzyme called UGGT (for UDP-glucose:glycoprotein glucosyltransferase) will add the glucose residue back onto the oligosaccharide thus regenerating the glycoprotein's ability to bind to calnexin. Glucosidase II can also remove the third and last glucose residue. These monoglucosylated oligosaccharides result from the trimming of two glucose residues by the sequential action of two glucosidases, I and II. Ĭalnexin binds only those N- glycoproteins that have GlcNAc2Man9Glc1 oligosaccharides. It specifically acts to retain unfolded or unassembled N-linked glycoproteins in the ER. Function Ĭalnexin is a chaperone, characterized by assisting protein folding and quality control, ensuring that only properly folded and assembled proteins proceed further along the secretory pathway. It consists of a large (50 kDa) N-terminal calcium- binding lumenal domain, a single transmembrane helix and a short (90 residues), acidic cytoplasmic tail. interleukin-35-mediated signaling pathwayĬalnexin (CNX) is a 67kDa integral protein (that appears variously as a 90kDa, 80kDa, or 75kDa band on western blotting depending on the source of the antibody) of the endoplasmic reticulum (ER).interleukin-27-mediated signaling pathway. 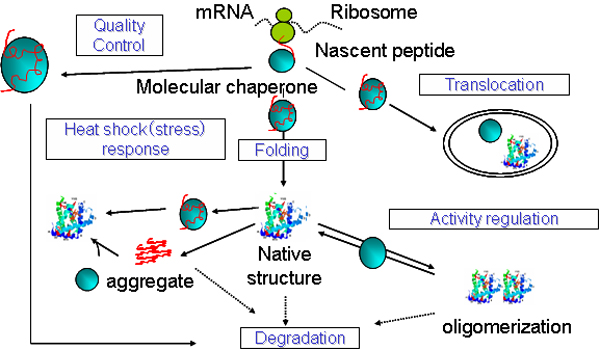
interleukin-12-mediated signaling pathway.protein folding in endoplasmic reticulum.antigen processing and presentation of peptide antigen via MHC class I.antigen processing and presentation of exogenous peptide antigen via MHC class II.Thus the concept of molecular chaperones does not contradict the principle of protein self-assembly, but qualifies it by suggesting that in vivo self-assembly requires assistance by other protein molecules. Known molecular chaperones do not convey steric information essential for correct assembly, but appear to act by binding to interactive protein surfaces that are transiently exposed during various cellular processes this binding inhibits incorrect interactions that may otherwise produce non-functional structures. The term assembly in this definition embraces not only the folding of newly synthesized polypeptides and any association into oligomers that may occur, but also includes any changes in the degree of either folding or association that may take place when proteins carry out their functions, are transported across membranes, or are repaired or destroyed after stresses such as heat shock. Molecular chaperones are currently defined in functional terms as a class of unrelated families of protein that assist the correct non-covalent assembly of other polypeptide-containing structures in vivo, but which are not components of these assembled structures when they are performing their normal biological functions. This introductory article proposes a conceptual framework in which to consider the information that is emerging about the proteins called molecular chaperones, and suggests some definitions that may be useful in this new field of biochemistry. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
